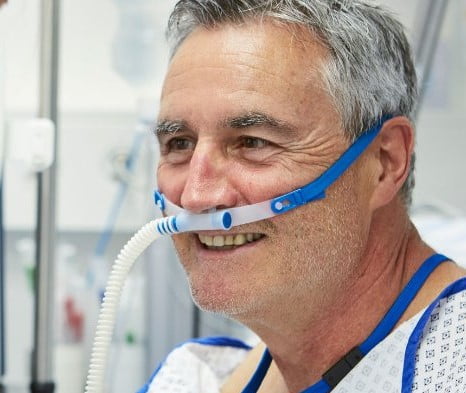
While not yet ready for field application, the high-flow nasal cannula is widely used in clinical settings to treat patients with hypoxemic respiratory failure. (Photo courtesy of Hamilton Medical)
The COVID pandemic has focused new attention on the value of the high-flow nasal cannula (HFNC). Early recognition by critical care practitioners that the ARDS-like presentation seen in critically ill COVID patients responded equally as well, if not better, to high concentration oxygen as it did to mechanical ventilation carved a niche for HFNC therapy in COVID care. High-flow nasal cannula refers to heated and humidified oxygen, delivered through a wide bore nasal cannula at flow rates of up to 60 liters per minute (LPM).
Related: High-Flow Nasal Cannula: It’s Not Just a Bunch of Hot Air!
The flow rates make HFNC a form of non-invasive ventilation (NIV). Without a tremendous oxygen supply, a HFNC setup would be difficult at best in the prehospital or critical care transport environments, but there are prototypes in development. Knowledge of this form of respiratory support is important for any emergency or critical care provider.
In most situations, prehospital and in-hospital, oxygen is typically delivered using low-flow devices like the nasal cannula. Occasionally, we need to escalate to use of high flow systems such as a venturi or non-rebreather mask. High-flow nasal cannula therapy utilizes a soft, large bore nasal prongs (cannula), humidification, heating and a gas blender. Heating and humidification are accomplished by placing a heater/humidifier similar to those used on mechanical ventilator circuits in line with the HFNC.1
The delivered gas, a blended mixture of compressed air and oxygen, set from 0.21 to 1.0 FiO2, runs through the heater/humidifier where it is warmed to body temperature and humidified to approximately the same 80-90% humidification seen physiologically with air inspired through the mouth and nose. The heating and humidification overcome the leading causes of discomfort seen with conventional oxygen therapy.2 The added moisture can help to liquefy mucus and facilitate expectoration.
The high flow washes out nasopharyngeal dead space, improving ventilation and oxygenation. There is also a somewhat difficult to quantify level of PEEP generated by the high flow. Theoretically, every 10 LPM of flow generates 1 CWP of PEEP.3 This PEEP (or CPAP) effect also decreases work of breathing and improves oxygenation.
Related: Why Peep?
Based on the delivered effects of HFNC, you may correctly surmise that it’s primary application is treatment of hypoxemic respiratory failure. Initiation and titration of HFNC is as much an art as a science. Application should be done at maximum flow (40 – 60 LPM for adults, depending on the brand equipment in use, and up to 8 LPM for infants). FiO2 should initially be set at 1.0 (100%). Flow is adjusted for patient comfort and to target respiratory rates between 25 and 30 BPM in adults. Before weaning flow, FiO2 should be weaned to at least 0.4 (40%). Once that is done, the flow can be weaned, typically in 5 LPM increments, assessing the effects. Temperature of the circuit is usually set to normal body temperature (37°C or 98.6℉) but can also be adjusted up or down for comfort.4-5
It is possible to transport most HFNC assemblies but doing so can be complex. Most require AC power for the heater/humidifier and need a stable platform to assure the heater/humidifier remains upright. The very high flows of gas typically necessitate large cylinders of both oxygen and compressed air, both connected to the blender that allows adjustment of the FiO2. In most cases, trying to move a HFNC setup is neither practical nor recommended. With that in mind, it is important to think critically about how to meet the oxygen and flow requirements of a patient on HFNC that you might be asked to transport.
Combining a non-rebreather mask with a nasal cannula might be sufficient to match the flows and FiO2 being delivered by HFNC, especially when the HFNC settings are not maximized. Pulse oximetry is a necessity for continual monitoring. An important, sometimes overlooked consideration, is the PEEP effect. A patient on HFNC at 40 LPM is likely getting 4 CWP of PEEP from the gas flow. Switching to a non-rebreather might provide the same amount of oxygen. If the PEEP was serving to reduce work of breathing and improve oxygenation, the disappearance of those benefits might not become evident for 15-20 minutes, which is likely to happen during transport.
Related: Keep the PEEP with SOKET!
A more suitable replacement for HFNC is CPAP. CPAP (or BiPAP), delivered using a transport ventilator, allows adjustment of the FiO2 and provides a means for continuing some level of PEEP. Disposable CPAP devices can deliver equivalent PEEP but may not be able to deliver adequate FiO2, depending on the manufacturer. Disposable CPAP units entrain significant amount of atmospheric air in order to achieve the gas flows needed. This mixing of oxygen with room air lowers the delivered FiO2. Adding a nasal cannula under a disposable CPAP mask might be sufficient to provide an adequate FiO2.
While not yet ready for field application, HFNC is widely used in clinical settings to treat patients with hypoxemic respiratory failure. The non-invasive nature of HFNC improves patient comfort and safety. An understanding of the principles is imperative for EMS providers called to transport patients being treated with HFNC. Safely moving a patient on HFNC necessitates matching both the FiO2, the gas flow and the PEEP likely being provided by the HFNC assembly. Eventually, we will see HFNC setups suitable to prehospital use.
References
- Dysart K, Miller TL, Wolfson MR, Shaffer TH. Research in high flow therapy: mechanisms of action. Respir Med 2009; 103:1400.
- Sztrymf B, Messika J, Mayot T, et al. Impact of high-flow nasal cannula oxygen therapy on intensive care unit patients with acute respiratory failure: a prospective observational study. J Crit Care 2012; 27:324.e9.
- Groves N, Tobin A. High flow nasal oxygen generates positive airway pressure in adult volunteers. Aust Crit Care. 2007;20:126-31.
- Parke RL, Eccleston ML, McGuiness SP. The effects of flow on airway pressure during high-flow oxygen therapy. Respir Care. 2011;56:1151-5.
- Mauri T, Alban L, Turrini C, et al. Optimum support by high-flow nasal cannula in acute hypoxemic respiratory failure: effects of increasing flow rates. Intensive Care Med 2017; 43:1453.
- Rochwerg B, Granton D, Wang DX, et al. High flow nasal cannula compared with conventional oxygen therapy for acute hypoxemic respiratory failure: a systematic review and meta-analysis. Intensive Care Med 2019; 45:563.
Mike McEvoy, PhD, NRP, RN, CCRN, is the EMS coordinator for Saratoga County, New York, and the professional development coordinator for Clifton Park and Halfmoon Ambulance. He is a nurse clinician in the adult and pediatric cardiac surgery intensive care units at Albany Medical Center, where he also teaches critical care medicine. McEvoy is the chief medical officer and firefighter/paramedic for West Crescent Fire Department in Clifton, New York. He is also the chair of the EMS Section board of directors for the International Association of Fire Chiefs and a member of the New York State Governor’s EMS Advisory Council. He is a lead author of the textbook Critical Care Transport, the “Informed” Pocket References (Jones & Bartlett), and the American Academy of Pediatrics textbook Pediatric Education for Prehospital Professionals (PEPP).


Recent Comments