On the go? Listen to the article in the player below!
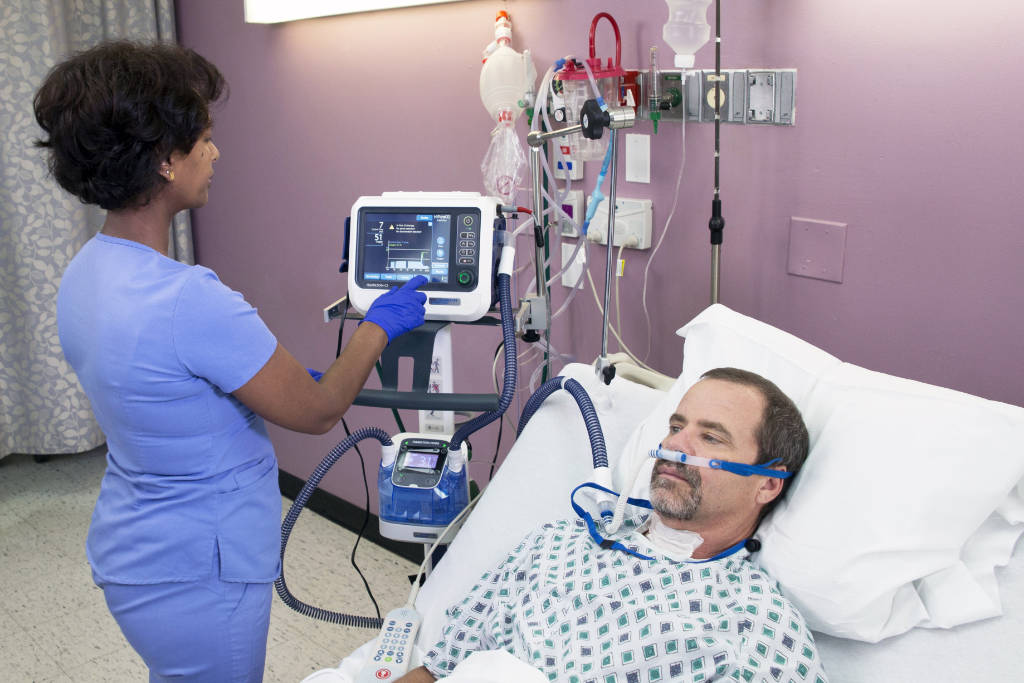
Not every patient experiencing a moderate level of hypoxemic respiratory failure does well on a high-flow nasal cannula. (Photo courtesy Hamilton Medical AG)When I first started as a hospital respiratory therapist, the nasal cannula was our go-to low-flow (LFNC) device, designed to deliver an FiO2 of approximately 24 to 44% at flows of 1 to 6 liters per minute. When patients needed more oxygen, we would typically consider a face mask using a humidified large volume nebulizer to deliver 35-50%. A tandem setup could deliver upward of 80-90%. When more oxygen was necessary, we had the NRB (non-rebreather) mask.
Around 1999, a device was developed with the ability to deliver flows of 40-60 LPM using a nasal cannula as the interface.1 That was not all it did. The device connected to an oxygen blender, allowing the practitioner to adjust the FiO2 from 0.21 all the way up to 1.0, and the delivered gas was heated and humidified. It seemed to be to be the greatest thing since nebulized Albuterol. At the time, it was so attractive that virtually every patient with acute shortness of breath ended up on the new high-flow nasal cannula (HFNC) device.
Still popular today, the flexibility of the HFNC allows for the successful treatment of patients with moderate levels of hypoxemic respiratory failure.2 Most patients prefer the nasal cannula interface over a face mask as they can speak, drink and eat while continuously wearing the nasal cannula. As a side note, it is not recommended that a patient on 60 LPM by HFNC be given a cheeseburger and a drink. There is a risk of aspiration at these high flow rates.
I, and my colleagues, eventually learned that not every patient experiencing a moderate level of hypoxemic respiratory failure does well on HFNC. It is important to consider what the patient’s cardio-respiratory status is and what other contributing factors may be involved.
You should then consider whether the patient needs oxygenation, ventilation or both. Is increased flow and FiO2 going to support or correct the patient’s situation? Or would positive pressure better support the patient’s respiratory needs? This is where your skills kick in, along with a quick history, including any known lung disease, to determine whether HFNC is appropriate.
Let’s discuss use of the HFNC in a hypothetical patient with COPD in moderate respiratory distress. The patient has an oxygen saturation of 83% on room air, and is now on a NRB at 15 LPM. Vital signs are HR 121, RR 28 and labored, BP 138/94, and the current sat is 90%. Breath sounds are diminished throughout. Given this limited information, it appears the patient is not oxygenating well. The NRB is likely supplying between 70 and 80% O2.
An ABG reveals partially compensated respiratory acidosis, confirming that the patient is not ventilating well. So, what is the best strategy for this patient? Because of the respiratory rate of 28 bpm and the hypoxemic and hypercapnic presentation, a trial of the HFNC would be a reasonable approach.
Because the HFNC can deliver an actual FiO2 of 1.0, at flows that will meet or exceed the patients inspiratory flow demand, it will allow for less atmospheric air to be entrained, contributing to a higher FiO2 in the lungs. The higher flows tend to wash out the CO2 from the upper airway, lowering anatomic dead space.3
Another advantage of the high flow rate is a decreased the work of breathing for the patient. Theoretically, the high flow rate results in a low level of CPAP being applied to the patient. While studies have produced data showing some amount of CPAP being applied to the lungs from HFNC flow rates, no method has yet been devised to actually measure these pressures.
There are also numerous confounding variables, including differences in pressures between the mouth being open or closed. Some studies suggest a pressure of about 3 CWP are generated at flows of 50 LPM. The investigators concluded that this generated CPAP pressure was of questionable clinical significance.4
Another important consideration is deciding whether the patient needs more flow or more pressure. In the case of our hypothetical COPD patient, HFNC is a reasonable place to start. Placing them on a flow of 40 to 60 LPM and FiO2 of 1.0, and titrating accordingly, would be typical. Accompany that with the usual cocktail of DuoNeb®, steroids and antibiotics, monitoring closely for improvement or decline.
What about a patient in respiratory distress who has a history of heart failure (HF)? Will high flow provide enough CPAP to help such a patient? Not likely. For suspected pulmonary edema, start patients on CPAP and move to Bi-Pap if needed.
What about a patient with severe respiratory hypoxemia from bilateral pulmonary embolisms? Is pressure added to the lungs going to help? Probably not, as this is a circulatory issue and the patient is ventilating dead space.
How about a cystic fibrosis patient in respiratory distress? Is this only an oxygenation issue, or do they also need CPAP/BIPAP? The key in making these decision is to go back to the question: Does the patient need assistance with oxygenation, ventilation, or both?
The major challenge for the transport industry is the ability to support high flows of blended medical air and oxygen for extended periods of time. By my calculations a patient on HFNC running at 40 LPM at an FiO2 of 1.0 would get 80 minutes of flow using a full M tank until the tank was completely empty (see What? We ran out of oxygen?). There are some manufacturers who recognize the need for HFNC during transport and are incorporating it into their ventilators.
The neonatal and pediatric patient populations may benefit from HFNC. I’ve witnessed thousands of kids benefit from HFNC from the NICU, PICU, and pediatric ED. Many neonates tolerate HFNC quite nicely and don’t have to be exposed to CPAP prongs or masks that may cause skin breakdown.
During RSV and bronchiolitis season, our university teaching hospital uses HFNC on a regular basis with good results, provided the patients come in early enough. Typical flow rates are 1-2 LPM/kg of IBW with titration of the FiO2 as needed. Because the pediatric population has a lower flow demand, it would be possible to use the HFNC in the field.
I believe early application of HFNC in the appropriate patient population is an ideal way to meet the patient’s inspiratory flow demand. Minimal training and education would be needed to implement this in the transport arena. Until field units are readily available, remember that it is all about oxygenation and ventilation, deciding what your patient needs, and your ability to apply this knowledge safely and effectively.
References
1. Cheifetz, I., MacIntyre, N. R., & Marini, J. J. (2017). Mechanical ventilation: essentials for current adult and pediatric practice. Mount Prospect, IL: Society of Critical Care Medicine, The Intensive Care Professionals.
2. Chatburn, R. L., El-khatib, M. F., & Volsko, T. A. (2016). Equipment for respiratory care. Burlington: Jones and Bartlett Learning.
3. Cheifetz, I., MacIntyre, N. R., & Marini, J. J. (2017). Mechanical ventilation: essentials for current adult and pediatric practice. Mount Prospect, IL: Society of Critical Care Medicine, The Intensive Care Professionals.
4. Hess, D., MacIntyre, N. R., Galvin, W. F., & Mishoe, S. C. (2020). Respiratory care: principles and practice. Burlington, MA: Jones & Bartlett Learning.
Michael Schauf, RRT-NPS, has been a registered respiratory therapist at Albany Medical Center Hospital in New York since 1994. He covers all in-patient units, ICUs, the adult and pediatric Emergency Departments and is a member of the neonatal/pediatric transport team at this Level I Trauma and Level 3 NICU Center. Michael is the founder of Vent-Pro Training, teaching medics and nurses around the world how to manage mechanically ventilated patients during ground, rotor, and fixed-wing transports. He has been a flight respiratory therapist since 1998, currently working for AirMed and as a clinical education specialist for AMR. Michael authored the respiratory chapter of, “Critical Care Transport, 2nd edition,” published by Jones & Bartlett and is an adjunct instructor in the Respiratory Care Program at Hudson Valley Community College.


Good article very informative